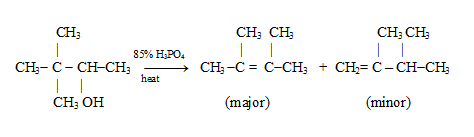General Methods of Preparation of Alkenes
Table of Content |
Dehydration of Alcohols
Heating most alcohols is a general method of preoarationof alkenes. Heating alcohols with a strong acid causes them to lose a molecule of water (to dehydrate) and form an alkene.
The reaction is an elimination and is favoured at higher temperatures. The most commonly used acids in the laboratory are Bronsted acids - proton donors such as sulphuric acid and phosphoric acid. Lewis acids such as alumina (Al2O3) are often used in industrial, gas phase dehydrations.
Dehydration reactions of alcohols show several important characteristics which shall be explained.
?The experimental conditions-temperature and acid concentration-that are required to bring about dehydration are closely related to the structure of the individual alcohol. Alcohols in which the hydroxyl group is attached to a primary carbon (primary alcohols) are the most difficult to dehydrate. Dehydration of ethanol, for example, requires concentrated sulphuric acid and a temperature of 180°C.
Secondary alcohols usually dehydrate under milder conditions. Cyclohexanol, for example, dehydrates in 85% phosphoric acid at 165-170°C.
Tertiary alcohols are usually so easily dehydrated that extremely mild conditions can be used, ter-butyl alcohol, for example, dehydrates in 25% H2SO4 at a temperature of 85°C.
Thus, overall, the relative ease with which alcohols undergo dehydration is in the following order
Ease of Dehydration: 3° Alcohol > 2° Alcohol > 1° Alcohol
This behaviour, is related to the stability of the carbocation formed in each reaction.
Some primary and secondary alcohols also undergo rearrangements of their carbon skeleton during dehydration.
Mechanism of Alcohol Dehydration : An E1 Reaction
The mechanism is an E1 reaction in which the substrate is a protonated alcohol (or an alkyloxonium ion). We consider the dehydration of CH3CHOHCH3 that proceeds through a carbonium ion intermediate. A catalytic role is assigned to the acid and O in ROH is a basic site.
Instead of HSO4-, a molecule of alcohol could act as a base in step 3 to give ROH2+.
Because step 2 is then, the rate determining step, it is the step that determines the reactivity of alcohols toward dehydration. The formation of a tertiary carbocation is easiest because the free energy of activation for step 2 of a reaction leading to a tertiary carbocation is lowest. The order of reactivity of the alcohols reflects the order of stability of the incipient carbonium ion (3°>2°>1°).
Carbocation Stability and the Occurrence of Molecular Rearrangements
Let us consider the rearrangement that occurs when 3,3-dimethyl-2-butanol is dehydrated
The first step of this dehydration is the formation of the protonated alcohol in the usual way. In the second step the protonated alcohol loses water and a secondary carbocation is formed. Next the less stable, secondary carbocation rearranges to a more stable tertiary carbocation.
The rearrangement occurs through the migration of an alkyl group (methyl) from the carbon adjacent to the one with positive charge. The methyl group migrates with its electron pair, i.e., as a methyl anion, -:CH3. After the migration is complete, the carbon atom that the methyl anion left has become a carbocation and the positive charge on the carbon atom to which it migrated becomes neutralized. Because a group migrates from one carbon to the next, this kind of rearrangement is often called a 1,2-shift.
The final step of the reaction, is the loss of proton from the new carbocation, and the formation of an alkene.
Path (b) leads to the highly stable tetrasubstituted alkene, and this path is followed by most of the carbocations. Path (a) leads to the less stable, disubstituted alkene and produces the minor product of the reaction.
The formation of the more stable alkene is the general rule (Saytzeff’s rule) in the acid-catalyzed dehydration of alcohols.
Reactions involving carbocations show that rearrangements are general phenomena. They occur almost invariably when the migration of an alkanide ion or hydride ion can lead to a more stable carbocation. The following are examples.
Rearrangements of carbocations can also lead to the change in ring size, as the following example shows:
Dehydrohalogenation of Monohalides
(a)Removal of HX from a substrate by alcoholic KOH or NaNH2
(b)This too is α–β elimination
(c)RCH2CH2X  RCH=CH2
RCH=CH2
CH3CH2CHCH3  CH3CH = CHCH3 + CH3CH2CH=CH2
CH3CH = CHCH3 + CH3CH2CH=CH2
X (Cf. Saytzeff rule)
(d)The case of dehydrohalogenation shows the order:
For alkyl halides: tertiary > secondary > primary alkyl halides
For halogens in halides : iodide > bromide > chloride
Dehalogenations of Dihalides
Vicinal (or vic) dihalides are dihalo compounds in which the halogens are situated on adjacent carbon atoms. The name geminal (or gem) dihalide is used for those dihalides where both halogen atoms are attached to the same carbon atom.Vic-dibromides undergo debromination when they are treated with a solution of sodium iodide in acetone or a mixture of Zn dust in acetic acid (or ethanol).

Debromination by sodium iodide takes place by an E2 mechanism similar to that for dehydrohalogenation.

Kolbe’s Electrolysis
Electrolysis of aqueous solutions of sodium or potassium salts of saturated dicarboxylic acids gives alkene.

Partial Hydrogenation of Alkynes
Hydrogenation of alkynes in presence of Lindlar catalyst gives alkenes.
RC≡CH  RCH=CH2
RCH=CH2
Lindlar catalyst is Pd on CaCO3 deactivated by lead acetate which prevents further hydrogenation.
Use of Pd or Pd-charcoal poisoned by BaSO4 and quinoline give better results. (Cram et. Al)
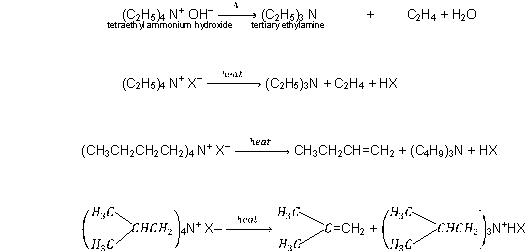
By Heating Quaternary Ammonium Compounds
By Grignard Reagents
(ix)Action of Copper alkyl on Vinyl Chlorides : Vinyl chloride on alkylation with copper alkyl form higher alkenes.
2H2C = CHCI  2H2C=CHR + CuCI2
2H2C=CHR + CuCI2
2H2C = CHCI  2H2C=CHR + CuCI2
2H2C=CHR + CuCI2
Related Sources
Refer the below mentioned links to get an immediate solution to all queries on Organic chemistry:
JEE Organic Chemistry Syllabus
View courses by askIITians


Design classes One-on-One in your own way with Top IITians/Medical Professionals
Click Here Know More

Complete Self Study Package designed by Industry Leading Experts
Click Here Know More

Live 1-1 coding classes to unleash the Creator in your Child
Click Here Know More


![CH_{3}-CH_{2}-OH\xrightarrow[180^{0}C]{conc. H_{2}SO_{4}}CH_{2}=CH_{2} +H_{2}O](https://files.askiitians.com/cdn1/cms-content/common/latex.codecogs.comgif.latexch_3-ch_2-oh_xrightarrow1800cconc.h_2so_4ch_2ch_2ampplush_2o.jpg)