Disaccharides
Table of Content |
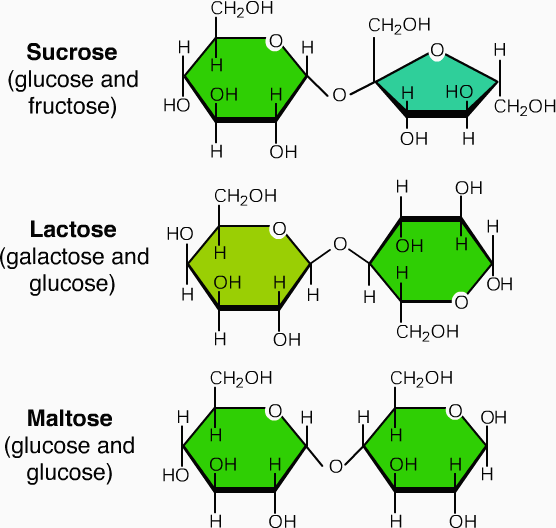
 Carbohydrates which upon hydrolysis give two molecules of the same or different monosaccharides are called disaccharides. Their general formula is C12H22O11. The three most important disaccharides are sucrose, maltose, and lactose. Each one of these on hydrolysis with either an acid or an enzyme gives two molecules of the same or different monosaccharides as shown below:
Carbohydrates which upon hydrolysis give two molecules of the same or different monosaccharides are called disaccharides. Their general formula is C12H22O11. The three most important disaccharides are sucrose, maltose, and lactose. Each one of these on hydrolysis with either an acid or an enzyme gives two molecules of the same or different monosaccharides as shown below:
Disaccharides may also be considered to be formed by a condensation reaction between two molecules of the same or different monosaccharides with the elimination of a molecule of water. This reaction involves the formation of an acetal from a hemiacetal and an alcohol – in which one of the monosaccharides acts as the hemiacetal while the other acts as the alcohol.
Now the question comes How do monosacharides combine to form disaccharides? The answer is through glycosidec bond or glycosydic linkage.
What is glycosidic linkage ?
Glycosidic linkage refers to the linkage formed between two monosaccharide units through an oxygen atom by the loss of a water molecule. For example maltose is formed by the combination two Glucose aunits. The C-1 and C-4 make glycosidic bond with elimination of water.
Maltose (C12H22O11)
Maltose or malt sugar is formed by condensation of two molecule of glucose joined with an α bond .Maltose is a reducing sugar. It reduces Tollens’ and Fehling’s reagent. It reacts with phenylhydrazine to yield hydrazone. It is oxidized by bromine water to maltobinoic acid. On hydrolysis it give two glucose units.
Maltose can be hydrolysed by enzyme maltase.
You can also refer to the following video for disaccharides
Sucrose (C12H22O11)

It is formed by condensation of one molecule of glucose and one molecule of fructose. Unlike maltose and lactose, it is non-reducing sugar since both glucose (C1 - α) and fructose (C2 -β) are connected to each other through their reducing centres. Its structure is shown below: Hydrolysis of sucrose ((Invert Sugar or Invertose) with hot dilute acid yields
D-glucose and D-fructose.
Sucrose is dextrorotatory, its specific rotation being +66.5%, D-glucose is also dextrorotatory, [α]D = +53°, but D-fructose has a large negative rotation, [α]D = -92°. Since D-fructose has a greater specific rotation than D-glucose, the resulting mixture is laevorotatory. Because of this the hydrolysis of sucrose is known as the inversion of sucrose, and the equimolecular mixture of glucose and fructose is known is invert sugar or invertose.
Lactose (C12H22O11)
Lactose or milk sugar is formed by condensation of one molecule of galactose and one molecule of glucos .Maltose is a reducing sugar. It reduces Tollens’ and Fehling’s reagent. It reacts with phenylhydrazine to yield hydrazone. Lactose exist in alpha and beta form which undergo mutarotation. It makes up about 5% of human milk and of cow’s milk. Milk sours when lactose in converted to lactic acids by bacterial (e.g. Lactobacillus bulgaricus). Acidic hydrolysis of lactose converts it into equal about of D- glucose and D- galactose.
Cellobiose (C12H22O11)
Cellobiose consists of two glucose molecules linked by a β bond. Octaacetate of cellobiose is obtained by treatement of cellulose with sulfuric acid. Alkaline hydrolysis of octaacetate yeilds cellobiose. Cellobiose is also a reducing sugar just like maltose. It forms osazone and exist in alpha and beta forms that undergi mutarotation. Cellobiose can be hydrolysed to two molecules of D-Glucose.Cellobiose differ from amtose in respect of enzyme which causes hydrolysis. Cellobiose is hydrolysed by emulsin and not be maltase.
Some other Related Links
JEE Organic Chemistry Syllabus
Reference books of Organic Chemistry
To read more, Buy study materials of Biomolecules comprising study notes, revision notes, video lectures, previous year solved questions etc. Also browse for more study materials on Chemistry here.
View courses by askIITians


Design classes One-on-One in your own way with Top IITians/Medical Professionals
Click Here Know More

Complete Self Study Package designed by Industry Leading Experts
Click Here Know More

Live 1-1 coding classes to unleash the Creator in your Child
Click Here Know More